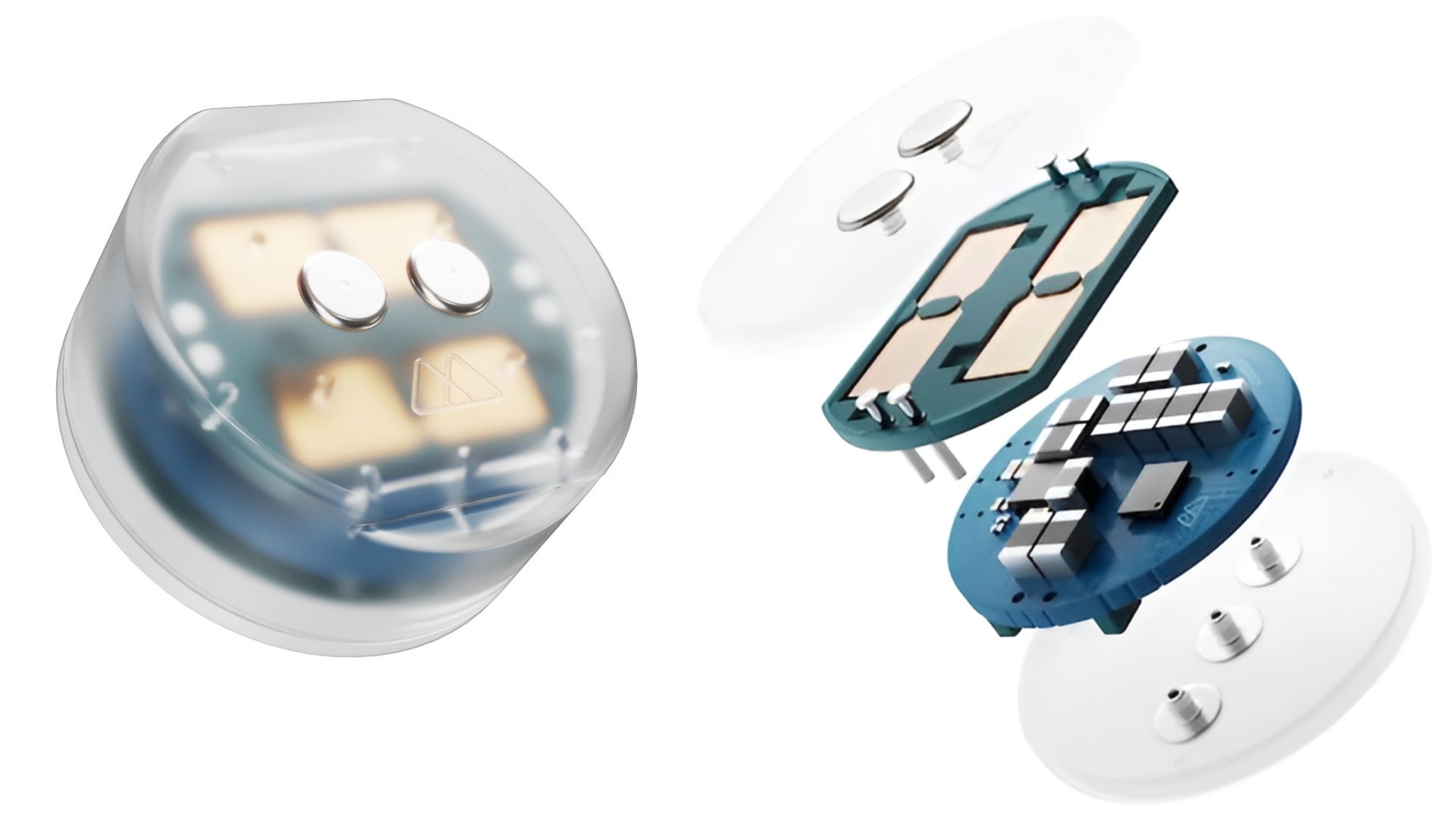
Treatment-resistant depression affects 3 million Americans, but Motif Neurotech’s DOT implant offers a wireless solution that feels straight out of a sci-fi thriller. The FDA just cleared the company’s investigational device for human trials—think of it as your brain getting its first real smart accessory. This isn’t some clunky medical contraption; it’s smaller than an AirPod and works without batteries or wires snaking through your skull.
The device targets patients who’ve failed multiple medications, offering precision neural stimulation through what CEO Jacob Robinson calls “a continuous glucose monitor for diabetes.” That comparison isn’t accidental—Motif wants you thinking wearable tech, not brain tissue surgery.
Installation: Easier Than Setting Up Your Router
The DOT implant sits on your skull bone without penetrating brain tissue, powered wirelessly from outside your head.
Here’s where things get interesting for the gadget-obsessed crowd. The installation takes 20 minutes in an outpatient setting—roughly the time needed to update your iPhone. The device sits on the dura (brain’s protective covering) without actually piercing brain tissue, making it less invasive than traditional deep brain stimulation.
Wireless power solves the biggest problem plaguing brain-computer interfaces: no batteries means no replacement surgeries, and no wires means dramatically reduced infection risk. Co-founder Sunil Sheth promises “an optimal balance of effectiveness and non-invasiveness,” which translates to getting brain stimulation without the typical surgical nightmare.
The device monitors brain signals continuously, adjusting stimulation patterns like your smartwatch tracking heart rate variability.
Trial Reality Check
The RESONATE trial launches at leading hospitals including Baylor College of Medicine and Massachusetts General Brigham.
Motif achieved FDA investigational clearance in four years—lightning speed for brain-computer interfaces, which typically take decades to reach human trials. The RESONATE study will track safety, device performance, and symptom reduction over 12 months across eight major medical centers.
This Rice University spinoff secured federal backing from NIH, DARPA, and ARPA-H, suggesting serious government interest in democratizing brain stimulation beyond current options. The technology stems from patented wireless power research, giving Motif a technical moat in an increasingly crowded neurotech space.
Whether this becomes the Fitbit of mental health or another overhyped medical gadget depends entirely on trial results. But the wireless approach finally makes brain stimulation feel like consumer technology rather than experimental medicine.